Discover our curated Epic Highlights, a testament to our commitment to healthcare excellence. Epic’s product portfolio ensures a seamless journey from Vial to Vein, offering advanced infusion solutions for every need. Explore the future of healthcare precision and quality.
Smart Mobile Apps

Revolutionizing infusion treatment beyond devices!
Epic SMART SOLUTIONS transcends conventional medical technology by delivering advanced mobile applications, such as eZVSCAN and SMARTeZ®Chemo CS, empowering users with cutting-edge solutions that go beyond mere infusion devices, to a modern patient treatment experience.



Pioneer Infusion Solutions
Epic R&D leads the way in the realm of infusion innovation.

World class manufacturer
Epic manufacturing facilities & processes ensure product quality and excess supply capacity.

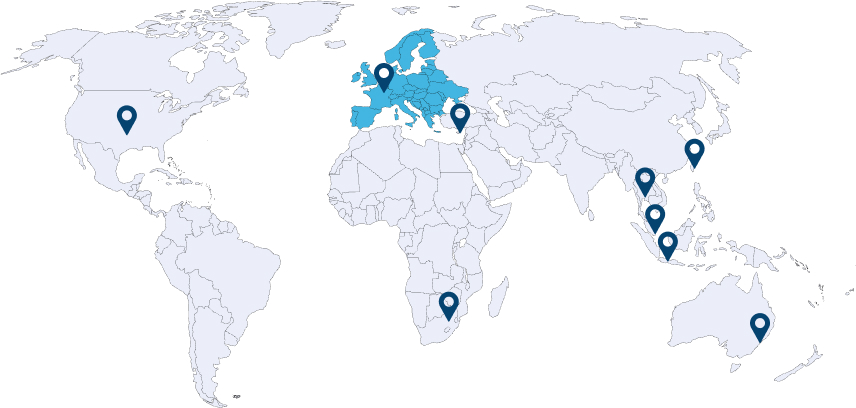
Globally Trusted Partner
EPIC products & services have garnered international recognition affirming their excellence in the global market..

From Vial to Vein!
EPIC solutions cater comprehensively to all all infusion needs, as in a One Stop Shop.
Since 2023, EPIC products are offered to the European Market by EPIC HEALTH EMEA
BECOME A PARTNER